Practice Fusion Perverted the Intent of Key Patient Safety Measures in Clinical Decision Support in its Electronic Health Record Software
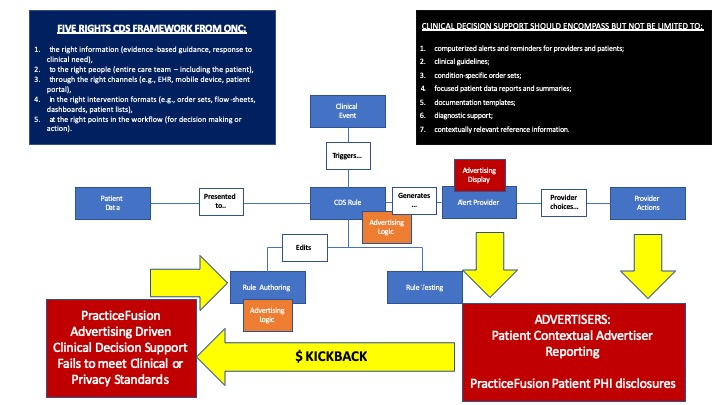
According to publicly available sources on the U.S. Department of Justice web site, in the U.S. v Practice Fusion case, opioid drug makers helped draft language fo so-called “clinical decision support” (CDS) alerts in its electronic health record (EHR) that included context-sensitive advertising for their opioid analgesics, and then paid kickbacks to ensure that Practice Fusion implemented them. Under the scheme, when a physician entered a certain phrase or diagnosis, the CDS pop up alert would suggest the drug maker’s product.
This case is a sad perversion of the intent of what Clinical Decision Support is supposed to do. The CDS requirements were designed to improve patient safety, not to encourage advertising and induce physicians to prescribe more pain killers.
I have personal experience in cases where preventable errors involving medication have nearly killed patients and permanently disabled them. A significant number of these cases involve opioids. I have also served as an expert in cases where hospitals have permanently disabled patients by failing to heed drug-drug and drug-allergy interaction warnings. These are some of the primary things that the Electronic Health Record Clinical Decision Support requirements were intended to safeguard against.
The Stage 2 Meaningful Use Final Rule states:
“CDS is not simply an alert, notification, or explicit care suggestion,” and goes on to describe non-alert CDS examples including disease-specific order sets and documentation forms/templates.” [1] The rule also defines Clinical Decision Support as “HIT functionality that builds upon the foundation of an EHR to provide persons involved in care processes with general and person-specific information, intelligently filtered and organized, at appropriate times, to enhance health and health care.”[2] [emphasis added].
But HHS did not intend that a CDS is simply an alert, notification, or explicit care suggestion. Electronic Health Records (aka Electronic Medical Records or EMRs) should encompass several components in its Clinical Decision Support capabilities.
CDS encompasses a variety of tools including, but not limited to:
-
computerized alerts and reminders for providers and patients;
-
clinical guidelines;
-
condition-specific order sets;
-
focused patient data reports and summaries;
-
documentation templates;
-
diagnostic support;
-
contextually relevant reference information.
According to the U.S. HHS Office of the National Coordinator (ONC), these functionalities may be installed on a variety of platforms, such as mobile, cloud, or installed,[3] but are “…NOT intended to replace clinician judgment, but rather [as] a tool to assist care team members in making timely, informed, higher quality decisions.”
ONC states that there are “Five Rights” that provide a best practice framework that providers may consider in considering Clinical Decision Support options appropriate for their practice. The Five Rights concept states that in order to provide these benefits,[4] CDS interventions must provide: [5], [6]
- the right information (evidence-based guidance, response to clinical need),
- to the right people (entire care team – including the patient),
- through the right channels (e.g., EHR, mobile device, patient portal),
- in the right intervention formats (e.g., order sets, flow-sheets, dashboards, patient lists),
- at the right points in the workflow (for decision making or action).
ONC continues, “CDS can be provided in various ways including, but not limited to, interruptive activities such as “pop-up” alerts, information displays or links (such as InfoButton), and targeted highlighting of relevant data. The key is that the information be presented when relevant, to those who can act on the information, and in a way that supports completion of the right action.” [emphasis added].
While many providers may associate CDS with pop-up alerts, alerts are not the only or necessarily the best method of providing support. For example, a pop-up alert can only fire *after* an event has occurred (e.g., a provider has ordered a contraindicated medication). [emphasis added]. One proven example of CDS is for abnormal blood pressure readings to automatically appear in red text (as opposed to normal blood pressure readings that appear in black) on providers’ displays. This method supports clinical workflow but does not interrupt the provider’s thought process or risk that an alert will be ignored due to ‘alert fatigue, ’ which has been identified as a key concern for implementers of CDS programs. [7]
Possible PracticeFusion HIPAA Breach
I have another concern with PracticeFusion’s scheme. The clinical decision support rules were based on patient specific clinical contexts. What data was shared with the advertisers who paid kickpacks? Was the patient data de-identified? Was Protected Health Information (PHI) disclosed by PracticeFusion?
[1] 77 FR 53997
[2] 75 FR 44350
[3] FDASIA Health IT report
[4] Osheroff, Teich, Levick et al., 2012. Improving outcomes with CDS: an implementer’s guide, Second Edition.
[5] AHIMA. The Five Rights of Clinical Decision Support: CDS Tools Helpful for Meeting Meaningful Use
[6] U.S. HHS Office of the National Coordinator HealthIT web site. “Clinical Decision Support: More than Just ‘Alerts’ Tipsheet” https://www.healthit.gov/sites/default/files/clinicaldecisionsupport_tipsheet.pdf
[7] “Alert fatigue” occurs when a provider, after receiving too many alerts or reminders (some of which may be irrelevant or unhelpful), begins to override or ignore further alerts without attending to them, thus potentially decreasing the care improvements expected from the tools (www.informatics-review.com/wiki/index.php/Alert_Fatigue).
Practice Fusion Advertising Funded CDSRelated Posts
Expert Witness Electronic Health Records